
Introduction
Engineers and product designers face a persistent challenge: aluminum delivers the lightweight efficiency and thermal performance modern applications demand, yet in its natural state, its surface remains vulnerable to corrosion, wear, and cosmetic degradation—particularly in demanding industrial environments. A heat sink exposed to salt spray, a chassis subjected to continuous handling, or a structural bracket cycling through temperature extremes will degrade quickly without reliable surface protection.
Anodizing transforms aluminum's surface at a molecular level into a harder, more durable, and functionally superior material. Unlike paint or plating, which sit on top of the metal, anodizing grows a dense aluminum oxide layer that is chemically bonded to the substrate itself—making it impossible to chip or peel off.
This guide breaks down the electrochemical process, the three main anodizing types, the material properties that matter for engineering decisions, and the real-world applications where anodized aluminum earns its place.
What Is Anodized Aluminum and How Is It Made?
Defining Anodized Aluminum
Anodized aluminum is aluminum that has undergone an electrochemical oxidation process to grow a thick, stable aluminum oxide (Al₂O₃) film on its surface — far thicker and more uniform than the thin native oxide that forms naturally in air. When exposed to atmosphere, pure aluminum self-passivates by forming an amorphous oxide layer only 2-3 nm thick.
By contrast, Type II sulfuric acid anodizing produces coatings of 1.8–25 μm (0.00007"–0.001"), while Type III hard anodizing can reach 13–150 μm (0.0005"–0.006"). That represents a thickness increase of 1,000 to 50,000 times over the natural oxide.
The Anodization Process
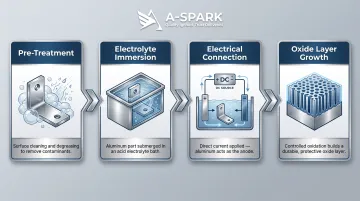
Anodization follows four defined steps:
- Pre-treatment: The aluminum part is cleaned and degreased to remove contaminants
- Electrolyte immersion: The part is submerged in an acidic electrolyte bath (typically sulfuric acid for Type II)
- Electrical connection: The aluminum component becomes the anode in a DC electrical circuit
- Oxide growth: When current flows, oxygen ions from the electrolyte react with the aluminum surface, growing the oxide layer both inward into the base metal and outward simultaneously
The Dual-Layer Structure
The resulting anodic oxide consists of two distinct layers:
- Barrier layer: A thin, non-porous layer at the aluminum-oxide interface
- Porous outer layer: A thicker oxide above the barrier, with nanopores 10–150 nm in diameter arranged in hexagonal arrays
This honeycomb-like columnar architecture is what enables dye absorption and sealing. The hexagonal pore model, first described by Keller et al. in 1953, explains the mechanism: as the oxide grows, the barrier layer reaction with the aluminum substrate drives pore formation in the outer layer.
Why the Oxide Cannot Peel
Because the anodized layer grows from within the base metal through a conversion reaction — not deposited on top of it — it cannot peel, flake, or chip. Unlike paint, powder coat, or plating, there is no interface between substrate and coating for adhesion to fail.
Sealing and Dyeing
After anodizing, the porous outer layer is sealed and optionally colored. The sequence works as follows:
- Dyeing (optional): Organic or metallic dyes are applied before sealing, producing vivid colors or stable bronze-to-black metallic tones
- Sealing: The part is immersed in hot deionized water or a chemical sealant (such as nickel acetate), closing the pores and completing corrosion protection
Key Properties of Anodized Aluminum
Surface Hardness and Abrasion Resistance
The aluminum oxide layer is significantly harder than base aluminum. Type III hardcoat anodizing produces surfaces approaching ceramic-level hardness—industry sources describe hardcoat as "harder than tool steel."
Hardness comparison data:
| Material | Hardness (HRC) | Hardness (HV) |
|---|---|---|
| Base 6061-T6 aluminum | ~15 HRC | Very soft |
| Type II anodized | 30-45 HRC | 200-400 HV |
| Type III hardcoat (optimized) | 50-60 HRC | 520-700 HV |
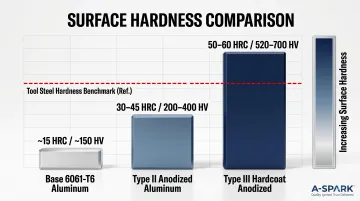
Note that hardness is alloy-dependent; some alloy-process combinations yield lower values. MIL-A-8625F specifies abrasion resistance via a Taber wear index: maximum 3.5 mg/1000 cycles for high-copper alloys, 1.5 mg/1000 cycles for others.
Corrosion Resistance
The dense oxide layer acts as a barrier against moisture, salt, and chemical attack — and the performance data backs this up.
AAMA 611 performance standards:
| Class | Thickness | Salt Spray (ASTM B117) | Application |
|---|---|---|---|
| Class I | 18 μm (0.7 mil) | 3,000 hours | Exterior curtain walls, coastal environments |
| Class II | 10 μm (0.4 mil) | 1,000 hours | Interior panels, light exterior use |
Class I coatings are specified for high-performance exterior architectural applications requiring long-term durability. Type III hardcoat separately exceeds 336 hours in 5% salt spray without corrosion signs.
Electrical Insulation
Anodized aluminum is non-conductive, making it a useful dielectric in electronic enclosures and insulating components. This property is especially valuable in EV battery enclosures operating above 800 volts, where anodizing provides essential insulation to mitigate short-circuit risk.
Thermal Properties
The anodized surface significantly affects both heat dissipation and emissivity. Anodized aluminum has higher emissivity than polished bare aluminum, making it more effective at radiating heat—a valuable property for heat sinks and thermal management components.
Emissivity transformation:
| Surface Condition | Emissivity |
|---|---|
| Polished aluminum | 0.03-0.06 |
| Anodized clear | 0.76-0.84 |
| Anodized black | 0.82-0.88 |

This represents a 20-30x increase in thermal radiation capability, making anodized aluminum a practical choice for passive thermal management in electronics enclosures, LED heat sinks, and aerospace thermal control systems.
Aesthetic and Coloring Versatility
Unlike paint or powder coat, color in anodized aluminum is locked into the oxide layer itself — making it resistant to peeling, chipping, and UV fade. The porous structure supports several coloring approaches:
- Organic dye impregnation: Vivid colors but variable lightfastness
- Electrolytic metallic salt deposition: Stable bronze-to-black tones
- Integral coloring: Color developed within the anodizing process itself
- Clear anodize: Retains the natural metallic silver-gray look
The Three Types of Anodizing
The MIL-A-8625 specification defines three internationally recognized anodizing types, each distinguished by electrolyte chemistry, process parameters, and resulting coating thickness — making them suitable for different engineering requirements.
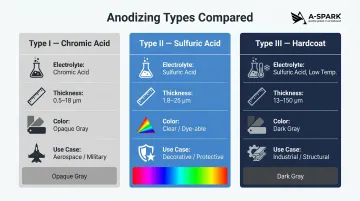
Type I – Chromic Acid Anodizing
Type I uses chromic acid as the electrolyte to produce very thin coatings (typically 0.03–0.10 mil or 0.5–18 μm). The resulting film is more opaque, slightly gray in color, and exhibits a dense, disordered microstructure. It offers excellent corrosion resistance with minimal dimensional impact on tight-tolerance parts.
Primary applications: Aerospace and military hardware where thin, uniform coatings are essential.
Regulatory concerns: Despite its performance, chromic acid anodizing faces significant restrictions. Chromium trioxide (CrO3) is listed on REACH Annex XIV (Entry 16) as a carcinogen, with a sunset date of September 21, 2017. Continued use in the EU requires formal authorization. This has pushed manufacturers toward Type IC alternatives using mineral or mixed mineral/organic acids.
Type II – Sulfuric Acid Anodizing
Type II is the most common commercial anodizing process. Using sulfuric acid electrolyte at controlled temperatures, it produces coatings from 0.1 to 0.7 mil (1.8–25 μm) in two sub-classes:
- Protective/Decorative (thinner): For lighting reflectors, appliance trim, automotive accents
- Architectural Class II (thicker): For interior panels and bleacher seating
Type II produces a clear, transparent coating that accepts dyes well, making it ideal for decorative and protective applications where color variety is desired.
Type III – Hard Anodizing (Hardcoat)
Type III uses lower temperatures (near freezing) and higher current densities (24–36 amps per square foot) to grow thicker, denser coatings (1.0–5.0 mil or 13–150 μm). Process voltages can reach 100 volts or higher.
- Rockwell C hardness in the 50–70 range under optimized conditions
- Exceptionally high abrasion and wear resistance
Primary applications: Machined parts, aircraft components, cookware, and hydraulic components.
Hardcoat finishes are often left unsealed to preserve maximum surface hardness, though this reduces corrosion resistance slightly compared to sealed coatings.
Benefits of Anodized Aluminum Over Untreated Aluminum
Durability Without Added Weight
Because the oxide layer grows from within the aluminum itself, anodizing adds minimal weight while extending service life by decades in demanding applications. Contrast this with applied coatings like paint or powder coat that add a surface layer and can fail adhesively over time. The oxide is integral to the substrate and cannot separate.
Environmental and Health Advantages
Anodizing is an electrochemical process with strong environmental credentials:
- Uses no VOCs (volatile organic compounds)
- Generates no EPA-classified hazardous waste in standard Type II applications
- Anodized aluminum is non-toxic and fully recyclable
- No intermediate processing needed to re-enter the recycling stream—unlike thicker organic or plated metallic coatings
Aluminum can be infinitely recycled without degradation of properties, and anodizing does not alter this recyclability. The recycling process saves approximately 95% of the energy required to produce aluminum from raw materials.
Long-Term Cost Efficiency
While anodizing adds upfront processing cost, it reduces total lifecycle cost by eliminating repainting, replating, and maintenance in corrosive or high-wear environments. AAMA 611 Class I coatings carry typical warranties of 5-10 years, with documented field performance of 15-30+ years in outdoor architectural applications when maintenance guidelines are followed.
Industrial Applications of Anodized Aluminum
Automotive and EV
Anodized aluminum is widely used in heat sinks, motor housings, battery enclosures, structural brackets, and exterior trim. Thermal performance, corrosion resistance, and a consistent surface finish make it practical for both structural and visible parts on the same vehicle platform.
EV-specific drivers:
- Provides dielectric insulation for battery enclosures operating above 800 volts
- Isolates high-voltage modules from conductive chassis components, reducing short-circuit risk
- Increases surface emissivity to improve heat dissipation in battery packs and power electronics
OEMs including BMW, Mercedes-Benz, and Volkswagen are driving demand for high-specification hard anodizing in structural and chassis components. The global aluminum anodizing market is projected at $2.7 billion in 2026, expected to reach $3.8 billion by 2033, growing at 5.0% CAGR, driven primarily by automotive lightweighting and EV component demand.

A-SPARK's aluminum die casting capabilities and IATF 16949-certified facilities support automotive and EV clients sourcing anodized components where electrical insulation and dimensional consistency are hard requirements.
Electronics and Consumer Devices
Laptop and smartphone chassis, LED lighting heat sinks, camera bodies, and electronic enclosures rely on anodized aluminum for scratch resistance, thermal dissipation, and premium aesthetics.
Notable examples:
- Samsung's Galaxy Alpha (2014) used color anodizing to tint metal frames in blue, gold, black, silver, or white
- Apple's MacBook line uses anodized aluminum unibody enclosures, with recent models incorporating up to 90% recycled aluminum
The anodizing process enables both the aesthetic finish and the scratch/corrosion resistance demanded in premium consumer devices.
Aerospace, Industrial Equipment, and Other Sectors
Hardcoat anodized aluminum is specified across sectors where surface hardness and chemical resistance are non-negotiable. Common applications include:
- Aircraft structural components and hydraulic pistons requiring tight dimensional tolerances post-anodizing
- Marine hardware and cookware exposed to corrosive environments
- Oil & gas equipment where chemical resistance and hardness prevent premature wear
In aerospace and oil & gas specifically, full traceability from raw material through surface treatment is a contractual requirement, not just a best practice. A-SPARK's in-house anodizing line, backed by ISO 9001:2015 and IATF 16949 certifications, covers the documentation chain — dimensional reports, process flow charts, quality control plans, and finishing certifications — that these regulated industries require.
Frequently Asked Questions
What does it mean for a metal to be anodic?
"Anodic" refers to the anode in an electrochemical cell—the electrode where oxidation occurs. When a metal is made anodic, it gives up electrons and reacts with oxygen ions. In aluminum's case, this forms a stable oxide layer rather than degrading the metal.
Is aluminum anodic?
Yes, aluminum is used as the anode in the anodizing process. Aluminum naturally tends to form a passive oxide layer in air—it is inherently inclined toward oxidation. Anodizing simply accelerates and controls this tendency to build a much thicker, engineered oxide film.
What is anodic alumina?
Anodic alumina (also called anodic aluminum oxide, or AAO) is the aluminum oxide material produced through anodization. It forms a self-organized nanoporous structure used both as a protective industrial finish and, in high-purity membrane form, as a nanotechnology platform.
What are the three types of anodizing?
Type I (chromic acid, thin, used in aerospace), Type II (sulfuric acid, the most common commercial process for decorative and protective finishes), and Type III (hardcoat, thick and wear-resistant, used in industrial and structural applications). Each type is defined by electrolyte chemistry, process parameters, and coating thickness.
How long does anodized aluminum last?
Properly sealed anodized aluminum—especially Architectural Class I and Type III hardcoat—can last 20-30+ years in outdoor or industrial environments, depending on coating thickness, alloy selection, and sealing quality. This longevity is well-documented in architectural field applications.
What are the limitations of anodized aluminum?
Anodizing doesn't work well on all alloys—high-silicon die-cast grades (such as A380 or ADC12) produce gray, porous films because silicon doesn't participate in the anodization reaction. The oxide layer also dissolves below pH 4 and above pH 10, limiting use in strongly acidic or alkaline environments, and hardcoat finishes can introduce minor dimensional changes to tight-tolerance parts.